 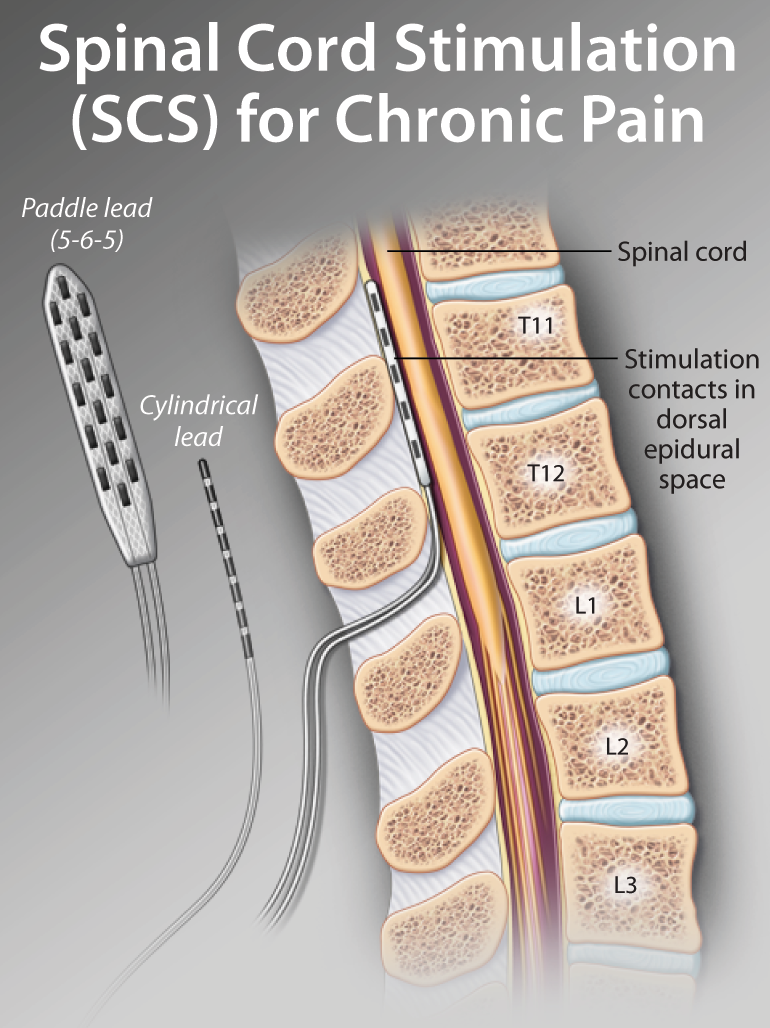
Subjects with CLBP reported this at 12, 18 and 24 months. Two-year outcomes comparing DTM SCS waveform to CMM saw superior back pain responder rates for Medtronic’s therapy. “Could closed-loop SCS eventually become the standard of care? More data is needed, but what we are seeing thus far is encouraging.” Medtronic has positive two-year results for DTM SCS Marc Russo, director of Hunter Pain Specialists in Newcastle, Australia and lead investigator. “These early promising results suggest that closed-loop SCS may offer real benefits over fixed-output stimulation,” said Dr. Morphine milligram equivalent reduced from 73.5 to 54.0. The study did not include a weaning protocol but 35% (37) of subjects on opioids at baseline reduced/stopped use at three months. This occurred without fear of pain or therapy side effects. All subjects reported satisfaction with closed-loop SCS therapy at three months.Īt three months more than 80% (54) of subjects achieved their stated activity goals for daily living with SCS therapy. Medtronic also reported that the study met its secondary objective, with 86% (51 subjects) reporting 50% or more reduction in overall back and leg pain at three months. Subjects reported clinically meaningful improvements in pain, physical function and quality of life at three months in closed-loop. Additionally, 86% reported a preference for closed-loop therapy during blinded testing at one month. The study met its primary objective, with 89% (28 people) of subjects reporting a significant reduction in overstimulation relative to open-loop. It also looked at the overall patient experience with the next-generation system. Medtronic presented one- and three-month outcomes from an evaluation of the long-term performance of its closed-loop SCS system. 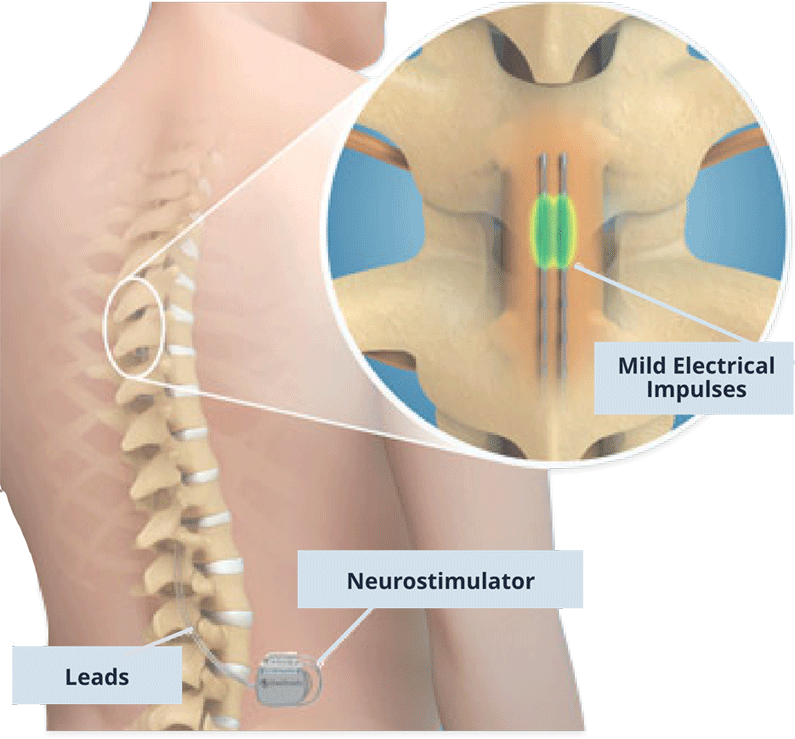
“We are committed to advancing the field of neuromodulation through rigorous scientific research in order to deliver on our Mission to alleviate pain, restore health and extend life to patients around the world.” Closed-loop SCS meets primary objective “Insights gained today from new clinical evidence pave the way toward the revolutionary therapies of tomorrow, and Medtronic is proud to be leading the way at NANS 2024 with 26 accepted clinical abstracts,” said Paolo Di Vincenzo, president of the Neuromodulation business at Medtronic. and Australia.Īdditionally, Medtronic shared 24-month outcomes for its DTM SCS compared to conventional medical management (CMM). The system holds European and Japanese approval but remains investigational in the U.S. That study looked at the company’s investigational, next-generation rechargeable SCS system. Medtronic’s data included evaluations of closed-loop SCS over three months in Australia. The company also plans to highlight its recently FDA-approved Percept RC deep brain stimulation system at NANS. Results demonstrated the benefits of SCS in patients with chronic low back pain (CLBP) and leg pain. The medtech giant shared data at the North American Neuromodulation Society (NANS) annual meeting in Las Vegas. + today announced data from two clinical trials supporting its spinal cord stimulation (SCS) technologies. Springer, Berlin Heidelberg New York Tokyo.The Vanta recharge-free neurostimulator offers DTM therapy. Zimmermann M (1975) Neurophysiological models for nociception, pain and pain trerapy. 
Shatin D, Mullet K, Hults G (1986) Totally implantable spinal cord stimulation for chronic pain: disain and efficiency. Richardson R, Signeira E, Cerullo L (1979) Spinal epidural neurostimulation for treatment of acute and chronic intractable pain: initial and long term results. Its diagnosis and treatment by spinal cord stimulation. Surg Neurol 4: 148–152ĭe La Porte Ch, Siegfried J (1983) Lumbosacral spinal fibrosis (spinal arachnoiditis). Nielson KD, Adams JE, Hosobuchi Y (1975) Experience with dorsal column stimulation for relief of chronic intractable pain. Nashold BS, Friedman H (1972) Dorsal column stimulation for control of pain. Halter J, Dolenc V, Dimitrijevic MR, Sharkey PC (1983) Neurophysiological assessment of electrode placement in the spinal cord. 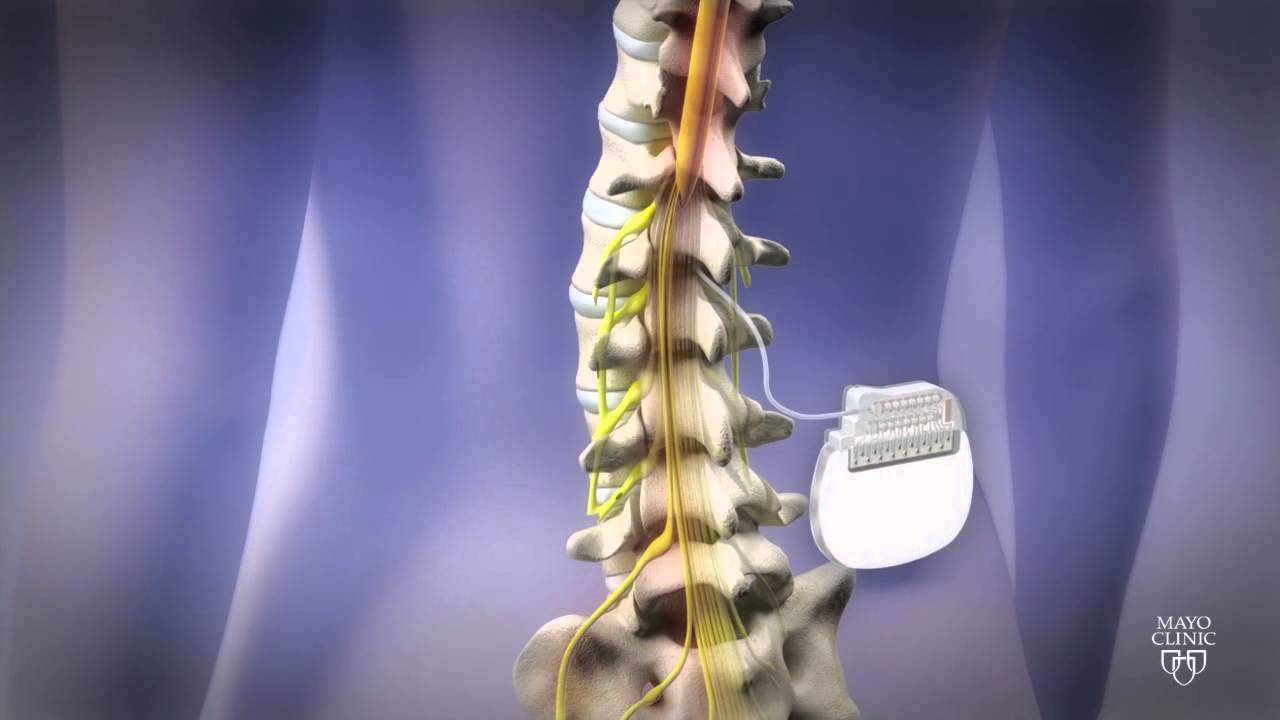
In Bonica JJ et al (eds) Advances in pain research and therapy. Appl Neurophysiol 46: 245–253Įrikseo DL, Long DM (1983) Ten years follow-up of dorsal column stimulation. Neurochirurgia 27: 47–50ĭimitrijevic MR, Faganet J, Sherwood AM (1983) Spinal cord stimulation as a tool for physiological research. Appl Neurophysiol 44: 160–170ĭemirel T, Braun W, Reimes CD (1984) Results of spinal cord stimulation in patients suffering from chronic pain a two year observation period. Davis R, Gray E (1981) Technical factors important to dorsal column stimulation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
